Biofourmis Selected by Vanderbilt University
BOSTON, February 16, 2022 – Biofourmis, a Boston-based global leader in virtual care and digital therapeutics, announced today that Vanderbilt University Medical Center has selected Biofourmis to support a clinical study measuring the safety and efficacy of monitoring patients in a home-like outpatient setting after receiving a leading oncology drug.
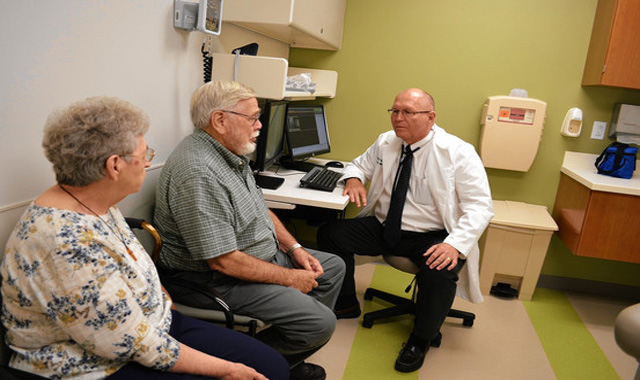
Patients with large B-cell lymphoma will receive the chimeric antigen receptor (CAR) T-cell therapy axicabtagene ciloleucel (YESCARTA®) by Kite, a Gilead Company. Instead of remaining in the hospital after receiving the immunotherapy, which is the current standard of care, patients will be discharged to a home-like setting near the medical center. In addition to periodic physical exams, several vital signs will be continuously monitored through Biofourmis’ analytics platform, which will assist the clinical team in detecting side effects of therapy to intervene as appropriate.
Patients receiving CAR-T therapy typically need to remain in the hospital up to seven to 10 days after the treatment, primarily to monitor for side effects, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). Both of these reactions can cause severe and potentially fatal side effects.
“CAR-T therapy is highly effective, but the lengthy hospital stays add cost to the system and inconvenience to patients,” said Olalekan O. Oluwole, MD, MPH, associate professor of Medicine, Hematology/Oncology at Vanderbilt University and primary investigator for the study. “Using remote patient management technology and telemedicine, we are investigating how patients can remain safely in a home-like setting while being monitored around the clock instead of in a hospital—and it is only those who develop specific symptoms and signs that will be admitted.”
Developing a New Standard of Care
In the study, patients’ heart rate, body surface temperature, oxygenation levels and respiratory rate will be monitored continuously while blood pressure will be collected every four to six hours. Biofourmis will deliver this collected data through a dashboard viewable by clinicians and researchers, who will also receive notifications based on vital signs when they deviate from baseline and/or if a study participant has not entered required blood pressure readings.
Biofourmis’ artificial intelligence (AI)-powered Biovitals® Platform, which received 510(k) clearance from the FDA in 2019 as a medical device for ambulatory physiological monitoring, continuously collects data from medical-grade biosensors to create a personalized view for each subject. This platform enables earlier detection of adverse events such as cytokine release syndrome (CRS) and neurotoxicity.
Clinicians may intervene based on data received through the Biofourmis platform. As an exploratory objective, Vanderbilt researchers will track the frequency of abnormal vital signs. The analytics will be used to identify trends in hospitalization and other outcomes.
“More and more, clinicians and researchers are exploring ways to safely and effectively care for patients from their homes, which is where most want to receive care for an acute condition or recovery after a hospital stay,” said Kuldeep Singh Rajput, CEO and founder of Biofourmis. “We are honored to partner with a highly respected research-driven organization such as Vanderbilt University Medical Center on this study utilizing Biofourmis’ technology to help clinicians detect early signs of an adverse reaction so they can deliver a preventive intervention. As a result, we believe this program fosters a new standard of care for the treatment of patients receiving CAR-T therapy.”
About Biofourmis
Biofourmis is a fast-growing global health technology company that is focused on leveraging software and data science to deliver virtual care and develop novel digital therapies. The company’s robust care management platform, Care@Home, enables remote disease management across a range of medical conditions for acute, post-acute and chronic care. The solution utilizes medical-grade wearables to continuously collect patient data, which is analyzed by Biovitals®, Biofourmis’ highly sophisticated, clinically validated AI-powered predictive analytics engine. With support from Biofourmis’ in-house clinical care team, payers and providers can leverage the solution to predict clinical deterioration in advance of a critical event—which enables earlier interventions for better outcomes and cost savings. Likewise, the company discovers, develops and delivers clinically validated digital therapeutics. These monotherapies or “pill plus” prescription therapeutics support payers and providers in improving patients’ lives while reducing healthcare utilization and associated costs. For more information, visit: www.biofourmis.com; and follow the company on LinkedIn and Twitter.
Media Contact:
Tara Stultz
Amendola Communications for Biofourmis
440.225.9595